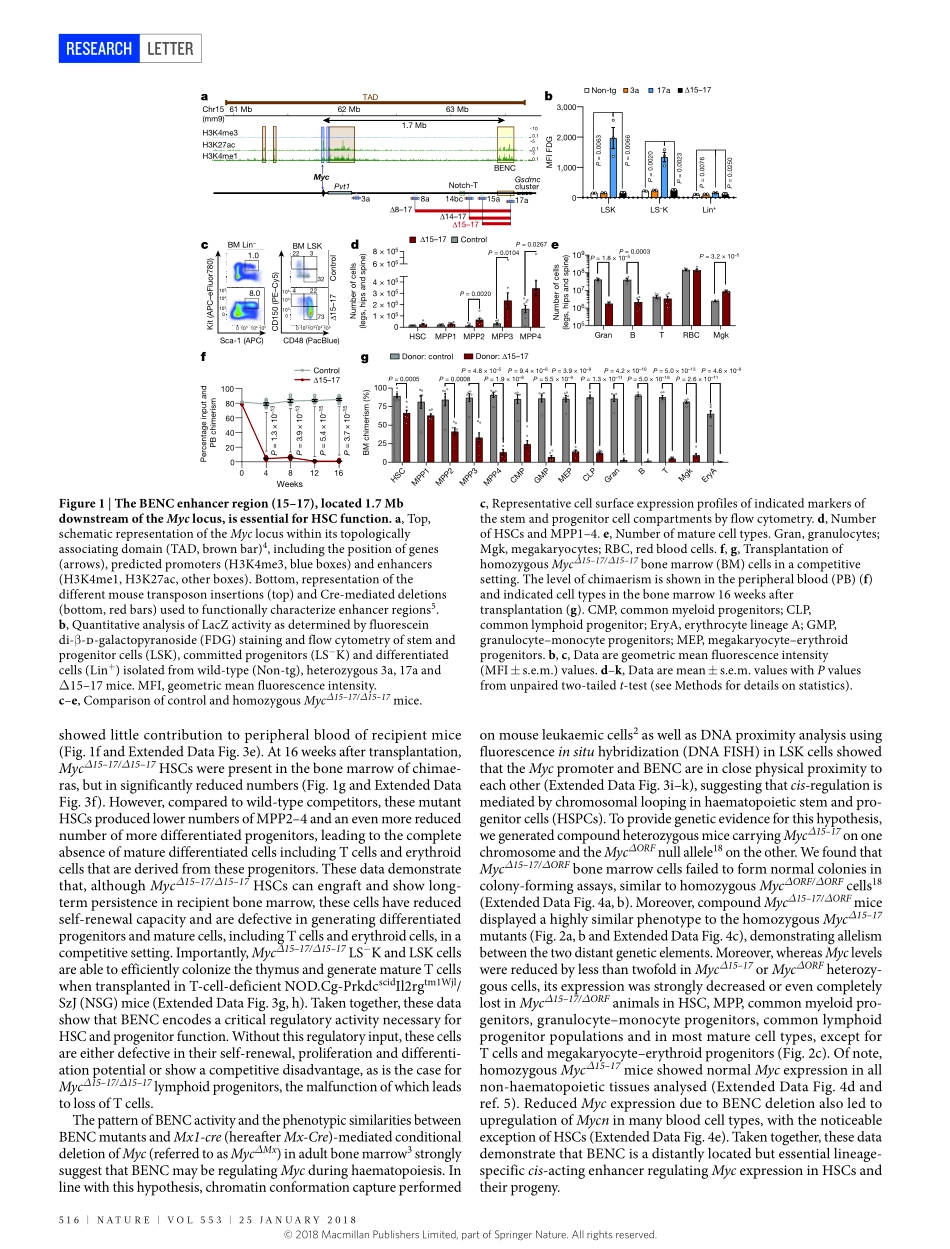
25january2018|VOL553|naTurE|515LETTErdoi:10.1038/nature25193AMycenhancerclusterregulatesnormalandleukaemichaematopoieticstemcellhierarchiesCarstenBahr1,2,3*,LisavonPaleske1,2,3*,VeliV.uslu4*,Silviaremeseiro4,naoyaTakayama5,6,StanleyW.ng7,alexMurison5,8,KatjaLangenfeld4,MassimoPetretich4,robertaScognamiglio1,2,3,PetraZeisberger1,2,amelieS.Benk1,2,3,Idoamit9,PeterW.Zandstra7†,MathieuLupien5,8,johnE.Dick5,6,andreasTrumpp1,2,3,10,11§&FrançoisSpitz4,12,13§ThetranscriptionfactorMycisessentialfortheregulationofhaematopoieticstemcellsandprogenitorsandhasacriticalfunctioninhaematopoieticmalignancies1.Hereweshowthatanevolutionarilyconservedregionlocated1.7megabasesdownstreamoftheMycgenethathaspreviouslybeenlabelledasa‘super-enhancer’2isessentialfortheregulationofMycexpressionlevelsinbothnormalhaematopoieticandleukaemicstemcellhierarchiesinmiceandhumans.DeletionofthisregioninmiceleadstoacompletelossofMycexpressioninhaematopoieticstemcellsandprogenitors.Thiscausedanaccumulationofdifferentiation-arrestedmultipotentprogenitorsandlossofmyeloidandBcells,mimickingthephenotypecausedbyMx1-Cre-mediatedconditionaldeletionoftheMycgeneinhaematopoieticstemcells3.Thissuper-enhancercomprisesmultipleenhancermoduleswithselectiveactivitythatrecruitsacompendiumoftranscriptionfactors,includingGFI1b,RUNX1andMYB.AnalysisofmicecarryingdeletionsofindividualenhancermodulessuggeststhatspecificMycexpressionlevelsthroughoutmostofthehaematopoietichierarchyarecontrolledbythecombinatorialandadditiveactivityofindividualenhancermodules,whichcollectivelyfunctionasa‘bloodenhancercluster’(BENC).WeshowthatBENCisalsoessentialforthemaintenanceofMLL–AF9-drivenleukaemiainmice.Furthermore,aBENCmodule,whichcontrolsMycexpressioninmousehaematopoieticstemcellsandprogenitors,showsincreasedchromatinaccessibilityinhumanacutemyeloidleukaemiastemcellscomparedtoblasts.ThisdifferencecorrelateswithMYCexpressionandpatientoutcome.Weproposethatclustersofenhancers,suchasBENC,formhighlycombinatorialsystemsthatallowprecisec...