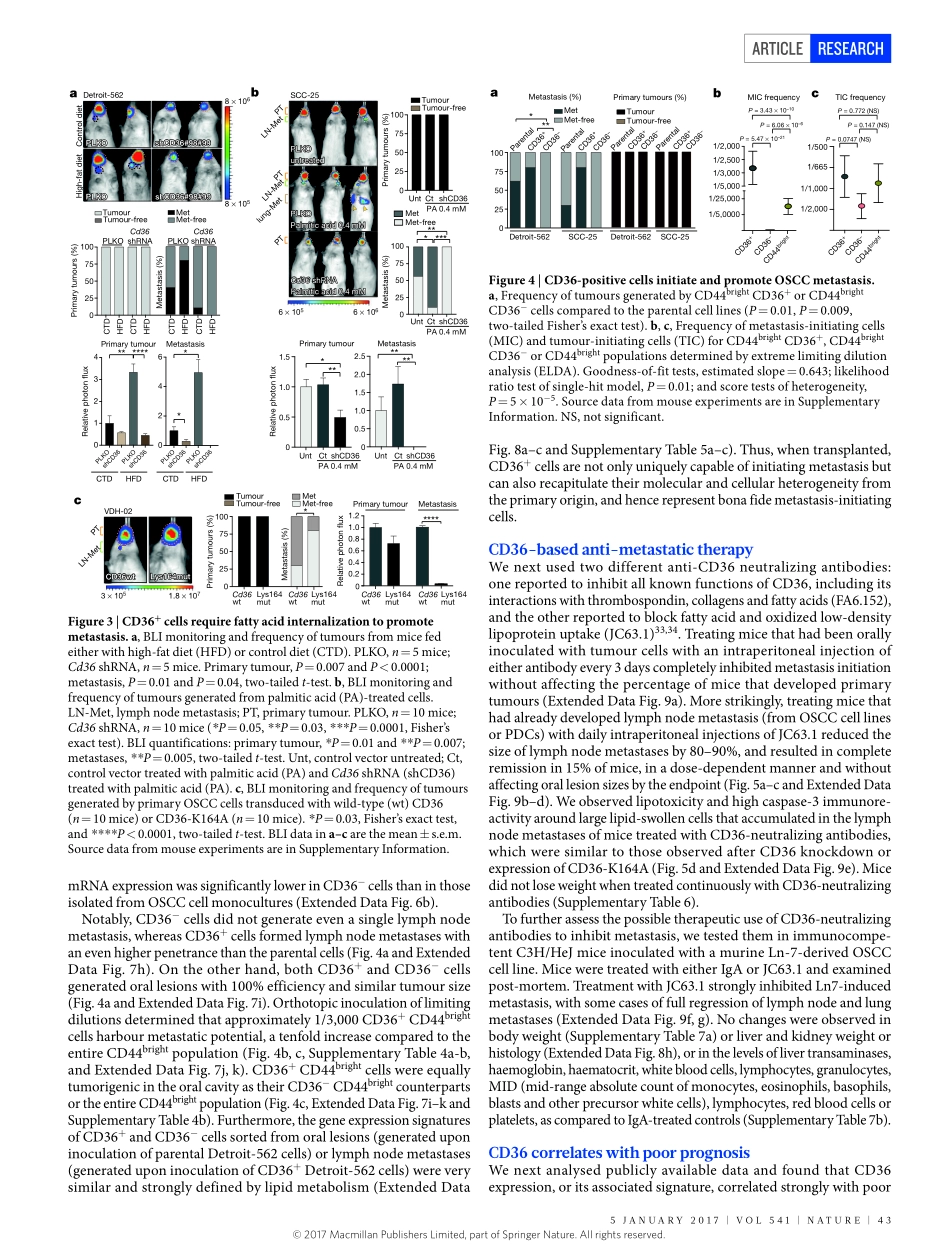
5january2017|VOL541|naTurE|41arTicLEdoi:10.1038/nature20791Targetingmetastasis-initiatingcellsthroughthefattyacidreceptorCD36GloriaPascual1,alexandraavgustinova1,StefaniaMejetta2,MercèMartín1,andréscastellanos1,camilleStephan-Ottoattolini1,antoniBerenguer1,neusPrats1,agustíToll3,juanantonioHueto4,coroBescós4,LucianoDicroce2,5,6&SalvadoraznarBenitah1,5Themechanismswherebysometumourcellsdetachfromtheprimarylesiontocolonizedistantsitesarestilllargelyunknown.Pro-metastaticeventscommontothemajorityofsolidtumoursmightincludethereversibletransitionoftumourcellsfromanepithelialtoamesen-chymalstateaswellastheirinteractionswithstromalcomponentsortumour-activatedstromalcells1–17.Sometumoursalsosecretemetastasis-promotingexosomesthatcontainproteins,mRNAsandmicroRNAstoestablishadistantpro-metastaticniche9,13,18,19.However,whetherasubpopulationofmetastasis-initiatingcellsexistsamongprimarytumour-initiatingcellsisnotclear.LRCsexpresslipidmetabolismgenesWhencelllinesandpatient-derivedcells(PDCs)arisingfromhumanoralcarcinomas(Methods)werepulsedwithalipophilicfluorescentdye(DiD)thatnon-specificallybindstomembranesandisdiluteduponcelldivision20,andwereorthotopicallyinjectedintotheoralcavityofNOD.Cg-PrkdcscidIl2rgtm1Wjl/SzJ(NSG)mice,weobservedasmallpercentageofslow-cyclingCD44bright/dye+long-termlabel-retainingcells(LRCs)withinorallesions(Fig.1a,bandExtendedDataFig.1a–k).Thus,theCD44brightpopulation,whichhavebeenshowntohavethehighesttumour-initiatingpotentialinoralsquamouscellcarcinomas(OSCCs),displayedcellcycleheterogeneityinvivo21–23.AlthoughthetranscriptomesofLRCs(CD44brightdye+)andnon-LRCs(dye−)sortedbyfluorescence-activatedcellsorting(FACS)fromorthotopictonguetumoursderivedfromtheOSCCcelllineSCC-25weremoresimilartoeachotherthantothedifferentiatedCD44dimpopulation,theystilldisplayedanumberofdifferentiallyexpressedgenes(ExtendedDataFig.2aandSupplementaryTable1a).GeneontologyanalysisindicatedthattheCD44brightdye−signaturewasassociatedwithchromosomalinstability,celltransf...