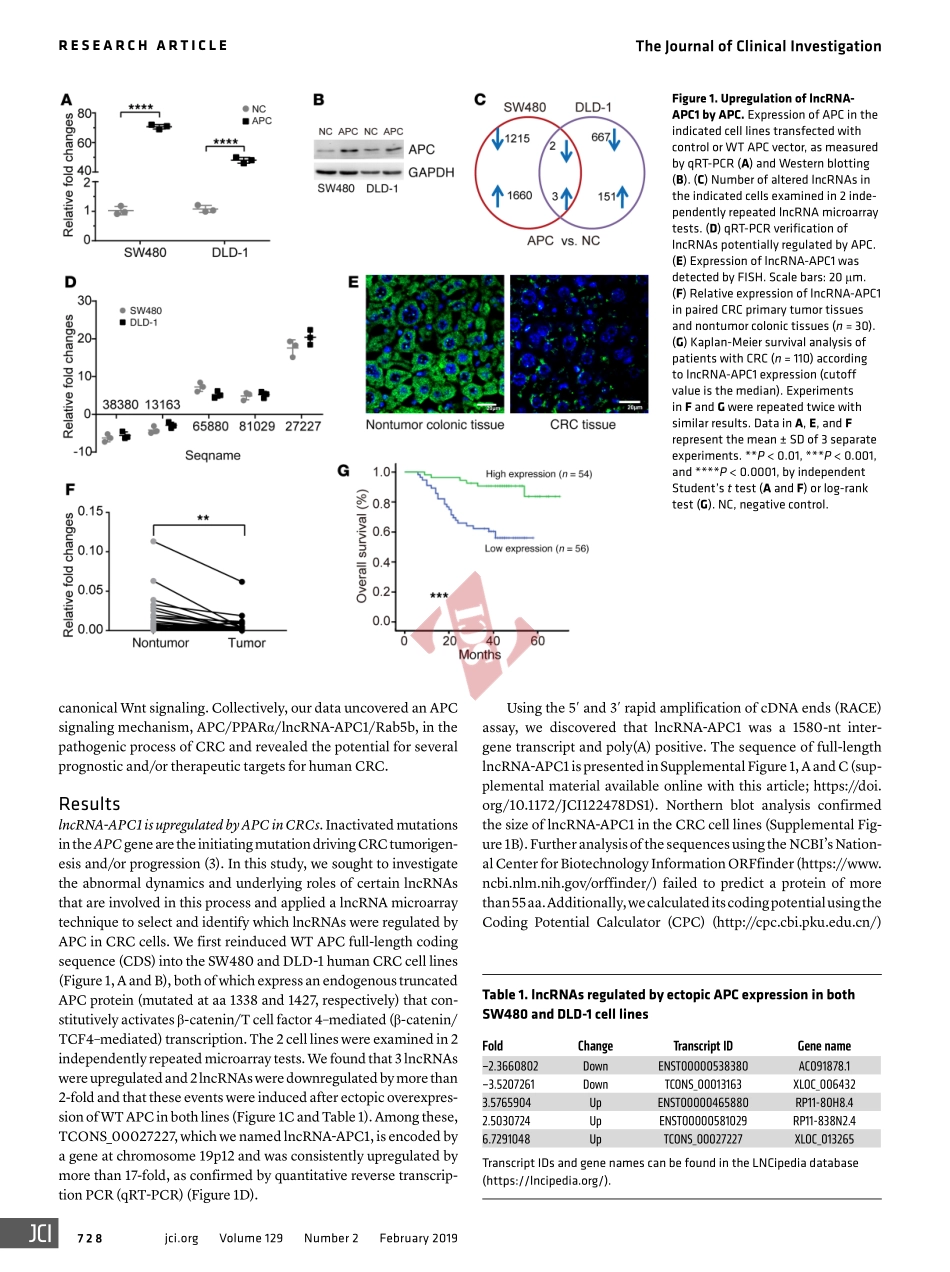
APC-activatedlongnoncodingRNAinhibitscolorectalcarcinomapathogenesisthroughreductionofexosomeproductionFeng-WeiWang,…,Rui-HuaXu,DanXieJClinInvest.2019;129(2):727-743.https://doi.org/10.1172/JCI122478.Theadenomatouspolyposiscoli(APC)geneplaysapivotalroleinthepathogenesisofcolorectalcarcinoma(CRC)butremainsachallengefordrugdevelopment.LongnoncodingRNAs(lncRNAs)areinvaluableinidentifyingcancerpathologiesandprovidingtherapeuticoptionsforpatientswithcancer.Here,weidentifiedalncRNA(lncRNA-APC1)activatedbyAPCthroughlncRNAmicroarrayscreeningandexamineditsexpressioninalargecohortofCRCtissues.AdecreaseinlncRNA-APC1expressionwaspositivelyassociatedwithlymphnodeand/ordistantmetastasis,amoreadvancedclinicalstage,aswellasapoorprognosisforpatientswithCRC.Additionally,APCcouldenhancelncRNA-APC1expressionbysuppressingtheenrichmentofPPARaonthelncRNA-APC1promoter.Furthermore,enforcedlncRNA-APC1expressionwassufficienttoinhibitCRCcellgrowth,metastasis,andtumorangiogenesisbysuppressingexosomeproductionthroughthedirectbindingofRab5bmRNAandareductionofitsstability.Importantly,exosomesderivedfromlncRNA-APC1–silencedCRCcellspromotedangiogenesisbyactivatingtheMAPKpathwayinendothelialcells,and,moreover,exosomalWnt1largelyenhancedCRCcellproliferationandmigrationthroughnoncanonicialWntsignaling.Collectively,lncRNA-APC1isacriticallncRNAregulatedbyAPCinthepathogenesisofCRC.OurfindingssuggestthatanAPC-regulatedlncRNA-APC1programisanexploitabletherapeuticapproachforthetreatmentofpatientswithCRC.ResearchArticleGastroenterologyOncologyFindthelatestversion:http://jci.me/122478/pdfTheJournalofClinicalInvestigationRESEARCHARTICLE727jci.orgVolume129Number2February2019IntroductionAsoneofthemostcommonhumanmalignancies,colorectalcar-cinoma(CRC)isaleadingcauseofcancer-relateddeathsworld-wide.ItiswellestablishedthatthepathogenesisofCRCfollowstheadenoma-carcinomasequenceandinvolvesmultisteptumor-igenesisthroughtheprogressiveaccumulationofabnormalitiesinbothtumorsuppressorgenesandoncogenes(1,2).Mutationsint...